Overview
Contraceptive injection
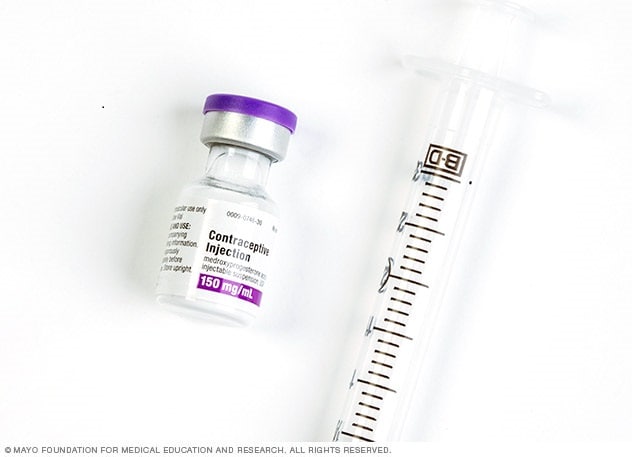
Contraceptive injection
The contraceptive injection contains the hormone progestin.
Depo-Provera is a well-known brand name for medroxyprogesterone acetate, a contraceptive injection that contains the hormone progestin. Depo-Provera is given as an injection every three months. Depo-Provera typically suppresses ovulation, keeping your ovaries from releasing an egg. It also thickens cervical mucus to keep sperm from reaching the egg.
Medroxyprogesterone acetate is also available in a lower dosage. This version is called Depo-SubQ Provera 104. While Depo-Provera is injected deep into the muscle, Depo-SubQ Provera 104 is injected just beneath the skin.
Depo-Provera and Depo-SubQ Provera 104 have similar benefits and risks. To use Depo-Provera or Depo-SubQ Provera 104, you'll need to visit your doctor or other health care provider.
Why it's done
Depo-Provera is used to prevent pregnancy and manage medical conditions related to your menstrual cycle. Your health care provider might recommend Depo-Provera if:
- You don't want to take a birth control pill every day
- You want or need to avoid using estrogen
- You have health problems such as anemia, seizures, sickle cell disease, endometriosis or uterine fibroids
Among various benefits, Depo-Provera:
- Doesn't require daily action
- Eliminates the need to interrupt sex for contraception
- Decreases menstrual cramps and pain
- Lessens menstrual blood flow, and in some cases stops menstruation
- Decreases the risk of endometrial cancer
Depo-Provera isn't appropriate for everyone, however. Your health care provider might discourage use of Depo-Provera if you have:
- Unexplained vaginal bleeding
- Breast cancer
- Liver disease
- Sensitivity to any component of Depo-Provera
- Risk factors for osteoporosis
- A history of depression
- A history of heart attack or stroke
In addition, tell your health care provider if you have diabetes, uncontrolled high blood pressure or a history of heart disease or stroke, and unexplained vaginal bleeding.
Risks
In a year of typical use, an estimated 6 out of 100 people using Depo-Provera will get pregnant. But the risk of pregnancy is much lower if you return every three months for your injection.
Depo-SubQ Provera 104 was highly effective in initial studies. However, it's a newer medication, so current research may not reflect pregnancy rates in typical use.
Among the things to consider about Depo-Provera are:
- You might have a delay in your return to fertility. After stopping Depo-Provera, it might take 10 months or more before you begin ovulating again. If you want to become pregnant in the next year or so, Depo-Provera might not be the right birth control method for you.
Depo-Provera doesn't protect against sexually transmitted infections. In fact, some studies suggest that hormonal contraceptives such as Depo-Provera might increase your risk of chlamydia and HIV. It isn't known whether this association is due to the hormone or behavioral issues related to the use of reliable contraception.
Using condoms will decrease your risk of a sexually transmitted infection. If you're concerned about HIV, talk with your health care provider.
It might affect bone mineral density. Research has suggested that Depo-Provera and Depo-SubQ Provera 104 might cause a loss of bone mineral density. This loss might be especially concerning in teens who haven't reached their peak bone mass. And it's not clear whether this loss is reversible.
Because of this, the U.S. Food and Drug Administration added strong warnings to the injection packaging cautioning that Depo-Provera and Depo-SubQ Provera 104 shouldn't be used for longer than two years. The warning also states that using these products might increase the risk of osteoporosis and bone fractures later in life.
If you have other risk factors for osteoporosis, such as a family history of bone loss and certain eating disorders, it's a good idea to discuss the potential risks and benefits of this form of contraception with your health care provider, as well as learn about other contraceptive options.
Other side effects of Depo-Provera usually decrease or stop within the first few months. They might include:
- Abdominal pain
- Bloating
- Decreased interest in sex
- Depression
- Dizziness
- Headaches
- Irregular periods and breakthrough bleeding
- Nervousness
- Weakness and fatigue
- Weight gain
Consult your health care provider as soon as possible if you have:
- Depression
- Heavy bleeding or concerns about your bleeding patterns
- Trouble breathing
- Pus, prolonged pain, redness, itching or bleeding at the injection site
- Severe lower abdominal pain
- A serious allergic reaction
- Other symptoms that concern you
Many experts believe progestin-only contraceptive methods, such as Depo-Provera, carry significantly lower risks of these types of complications than do contraceptive methods that contain both estrogen and progestin.
How you prepare
You'll need a prescription for Depo-Provera from your health care provider, who will likely review your medical history and possibly check your blood pressure before prescribing the drug.
Talk to your health care provider about all your medications, including nonprescription and herbal products. If you want to give yourself Depo-Provera injections at home, ask your health care provider if that's an option.
What you can expect
To use Depo-Provera:
Consult your health care provider about a starting date. To ensure you're not pregnant when you're injected with Depo-Provera, your health care provider will likely give you your first injection within seven days of the start of your period.
If you've just given birth, your first injection will be done within five days of giving birth, even if you're breast-feeding. You can start Depo-Provera at other times, but you might need to take a pregnancy test first.
Prepare for your injection. Your health care provider will clean the injection site with an alcohol pad. After the injection, don't massage the injection site.
Depending on your start date, your health care provider might recommend that you use a backup method of birth control for seven days after your first injection. Backup birth control isn't necessary after subsequent injections as long as they're given on schedule.
Schedule your next injection. Depo-Provera injections should be given every three months. If you wait longer than 13 weeks between injections, you might need to take a pregnancy test before your next injection.
Feb. 22, 2022