Overview
Polio is an illness caused by a virus that mainly affects nerves in the spinal cord or brain stem. In its most severe form, polio can lead to a person being unable to move certain limbs, also called paralysis. It can also lead to trouble breathing and sometimes death. The disease also is called poliomyelitis.
A vaccination effort throughout the world has led to only a small number of cases to occur around the world in recent years. But poliovirus still spreads within areas with low vaccination rates.
The U.S. Centers for Disease Control and Prevention (CDC) publishes travel notices of countries where there is a higher risk of polio. Countries at a higher risk of polio are generally in Africa, the Middle East, and southern and central Asia.
Vaccinated adults who plan to travel to an area where polio is spreading should get a booster dose of inactivated poliovirus vaccine (IPV). Immunity after a booster lasts a lifetime.
Symptoms
Nerve cell (neuron)
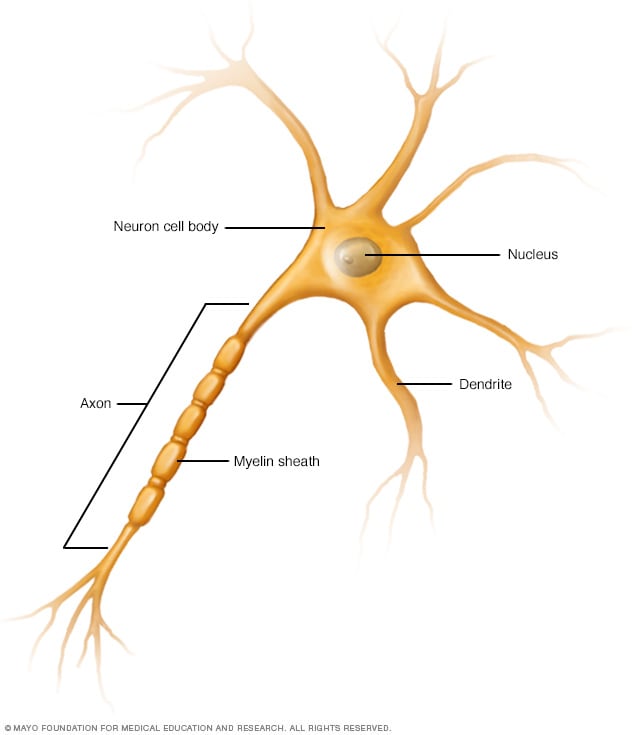
Nerve cell (neuron)
The basic unit of communication in the nervous system is the nerve cell (neuron). Each nerve cell consists of the cell body, which includes the nucleus, a major branching fiber (axon) and numerous smaller branching fibers (dendrites). The myelin sheath is fatty material that covers, insulates and protects nerves of the brain and spinal cord.
Most people infected with the virus that causes polio, called poliovirus, don't get symptoms.
Abortive polio
About 5% of people with the poliovirus get a mild version of the disease called abortive poliomyelitis. This leads to flu-like symptoms that last 2 to 3 days. These include:
- Fever
- Headache
- Muscle aches
- Sore throat
- Stomachache
- Loss of appetite
- Nausea
- Vomiting
Nonparalytic polio
A more severe form of the disease, called nonparalytic polio, affects about 1% of those infected. While the illness lasts longer than a few days, it doesn't cause paralysis. Besides having more-severe flu-like symptoms, nonparalytic polio symptoms may include:
- Neck pain or stiffness
- Aches or stiffness in the arms or legs
- Severe headache
A second phase of symptoms may follow, or a person may seem to be getting better for a few days before a second phase starts. These symptoms include:
- Stiffness of the spine and neck
- Decreased reflexes
- Muscle weakness
Paralytic polio
This most serious form of the disease is rare. The disease begins much like nonparalytic polio. But it progresses to more-severe signs and symptoms, including:
- Intense pain
- Extreme sensitivity to touch
- Tingling or pricking sensations
- Muscles spasms or twitching
- Muscles weakness progressing to a limp paralysis
Any combination of limbs may experience paralysis. But paralysis of one leg is most common, followed by paralysis of one arm.
Depending on the severity of disease, other signs or symptoms may include:
- Paralysis of muscles involved in breathing
- Difficulty swallowing
Post-polio syndrome
Post-polio syndrome is the appearance of new signs or symptoms or the progression of problems. This usually happens decades after having polio. Common signs and symptoms include:
- Progressive muscle or joint weakness and pain
- Fatigue
- Muscle wasting
- Breathing or swallowing problems
- Sleep-related breathing disorders, such as sleep apnea
- Lowered tolerance of cold temperatures
When to see a doctor
Signs and symptoms of polio are similar to other viral diseases that affect the nervous system. It's important to get a timely and accurate diagnosis.
If you had polio before, see your health care provider if you have new or worse signs or symptoms.
Causes
Polio is caused by the poliovirus. It mainly targets nerve cells in the spinal cord and brain stem that control muscle movement. Nerve cells controlling sensation are generally not affected.
The naturally-occurring poliovirus, called the wild-type poliovirus, has been eliminated in most countries and causes few cases of polio. Another version of the virus, called the vaccine-derived poliovirus (VDPV), is more widespread and now causes most infections worldwide. VDPV exists mainly in a few countries that use an oral vaccine with a weakened poliovirus.
The weakened virus in the oral vaccine doesn't itself cause polio, and vaccinated people rarely contract VDPV. Instead, VDPV is a new version of the virus that develops within a community or region where not enough people are vaccinated.
Even though the weakened virus in the oral vaccine doesn't cause illness, it can spread. If most people in a community are vaccinated, the spread of the weakened virus is controlled. If many people aren't vaccinated, the weakened virus can move through a community for a long time. This gives the virus the chance to change, or mutate, and behave like the wild-type virus that causes illness.
Infections from VDPV have been reported in the United States. In each case, the person was either not vaccinated or had a significantly weakened immune system. One case in New York in 2022 was in a county with a lower-than-average polio vaccination rate. Samples from wastewater showed that VDPV was spreading in some communities.
Since 2000, polio vaccination in the United States has used an injected vaccine with an inactivated poliovirus that doesn't create the risk for VDPV.
How polio spreads
People carrying the poliovirus — even people who don't get sick — can pass along the virus in feces, also called stool, or droplets from sneezing or coughing. The virus enters another person through the mouth. The virus can spread easily. For example, the virus can spread if people haven't washed their hands after coughing, using the toilet or before eating.
The virus also may be in water contaminated with feces carrying the poliovirus.
Risk factors
Polio mainly affects children. But anyone who hasn't been vaccinated is at risk of getting the disease.
Complications
Severe disease that affects the ability to breathe can cause death. Long-term complications for people who recover may include:
- Permanent paralysis
- Muscle shortening that causes deformed bones or joints
- Chronic pain
- Post-polio syndrome
Prevention
The most effective way to prevent polio is vaccination.
Polio vaccine
The CDC recommends four doses of inactivated poliovirus vaccine (IPV) at the following ages:
- 2 months
- 4 months
- Between 6 and 18 months
- Between ages 4 and 6 when children are just entering school
If your child is missing a dose, talk to your health care provider about a catch-up schedule for vaccination.
Adult vaccination
In the U.S., most adults have immunity to the poliovirus because of childhood vaccination. And U.S. adults have little chance of being exposed to the virus. But adults who are traveling to or living in a region with high rates of spreading poliovirus should receive more vaccinations.
If you had a complete course of vaccination, you should get a one-time IPV booster. You should get three IPV doses if you aren't vaccinated, didn't receive a complete vaccination or are unsure of your vaccination status.
The IPV schedule for adults is a second dose 1 to 2 months after the first dose. Then the third dose is 6 to 12 months after the second dose. Talk to your health care provider if you know you have an incomplete vaccination history.
Vaccine safety
IPV is safe for people with weakened immune systems, although it's not certain how protective the vaccine is in cases of severe immune deficiency. Common side effects are pain and redness at the injection site.
IPV can cause an allergic reaction in some people. Because the vaccine has trace amounts of the antibiotics streptomycin, polymyxin B and neomycin, it may cause a reaction in people allergic to one of these antibiotics. A person who has a severe reaction to a first dose of IPV won't get more doses.
Signs and symptoms of an allergic reaction usually occur within minutes to a few hours after the shot. Watch for:
- Skin reactions, including hives and itching and flushed or pale skin
- Low blood pressure (hypotension)
- Narrowing of the airways and a swollen tongue or throat, which can cause wheezing and trouble breathing
- A weak and fast pulse
- Nausea, vomiting or diarrhea
- Dizziness or fainting
If you or your child has an allergic reaction after any vaccination, get medical help right away.
Jan. 05, 2023